- Home
- Services
- About
- News
- Contact
- Shader model 3-0
- Gta 5 mod menu download xbox one
- Cool og gamertags not taken
- Hr xps peak pd 3d
- Combos de the king of fighters 2002 para maquinitas
- Dragon age inquisition patch 1-12 cheats
- Run 8 train simulator multiplayer servers
- Enochian chess set
- Spartacus all seasons watch online with english subtitles
- Rufus ubuntu
- Ems topgun 3 ps2
- Pharaoh game
- Hex color code soft navy blue
- Concorde c40 modem driver
- Flstudio video
- Convert pdf to cad autocad 2018
- Xbox 1 controller driver
- Pokemon soul silver rom nds file
- Price zimbra network edition
- Bescor mp-101 motorized pan tilt head
- Free mujhse dosti karoge movie online
- Myanmar xxx
- Re download os x el capitan
- Clear cookies and cache in firefox mac os x
- Cant upgrade to 64 bit windows 10
- Ibm i access client solutions screen flashes
- Novel dewasa online
- Mac website builder software
- Download film wiro sableng 2018 bluray
- Seagate file recovery for windows registration key free
- Best tamil gana songs mp3 free download
- Mac battery life short
- Convert mod to avi file
- Intel centrino advanced n 6235 driver windows 7 64 bit
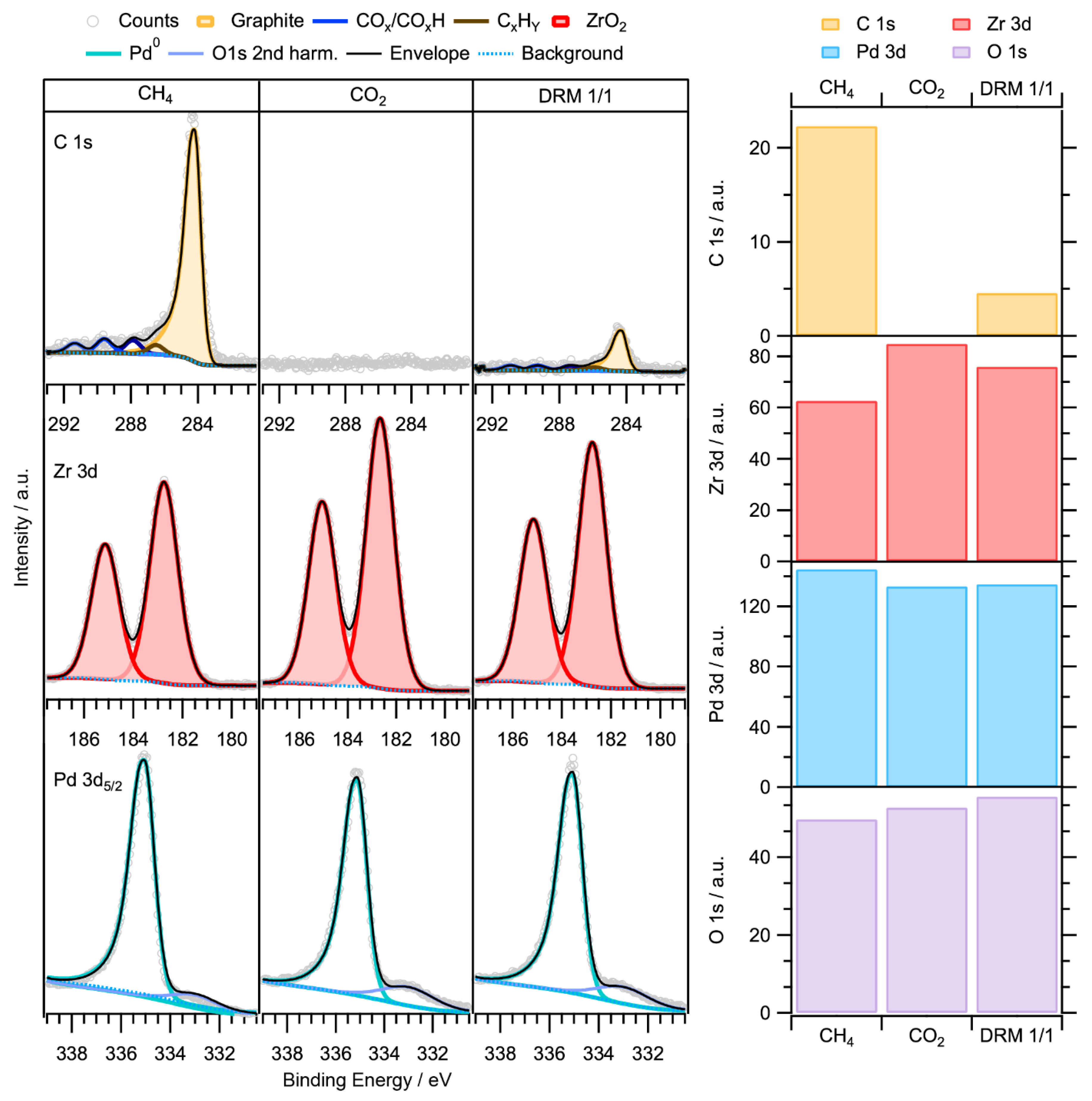
reported SMSI formation between Au NPs and TiO 2 induced by melamine under an oxidative atmosphere. One critical element in this pursuit is switching the high-temperature treatment in H 2 into other conditions and thereby changes the mechanistic pathways for the formation of SMSI. Efforts have been devoted in hope of expanding upon the conventional SMSI. Consequently, it is extremely challenging for some metals, e.g., Au, to manifest SMSI due to their low work function and surface energy 15, 17, 22. Under extreme conditions, the formation of intermetallic structure of the supported metal and metal cations in the supporting oxide was observed 20, 21.ĭespite these fascinating interfacial properties in SMSI, the formation of SMSI is restricted to specific combinations of elements, i.e., group VIII metals with high surface energy and transition metal oxides with low surface energy. Meanwhile, because the reducible oxide support, e.g., TiO 2, Co 3O 4, CeO 2, and Nb 2O 5, is partially reduced to the structure with a nonstoichiometric oxygen concentration during the reductive annealing, electron transfer between metal NPs and oxide supports was detected 16, 17, 18, 19.
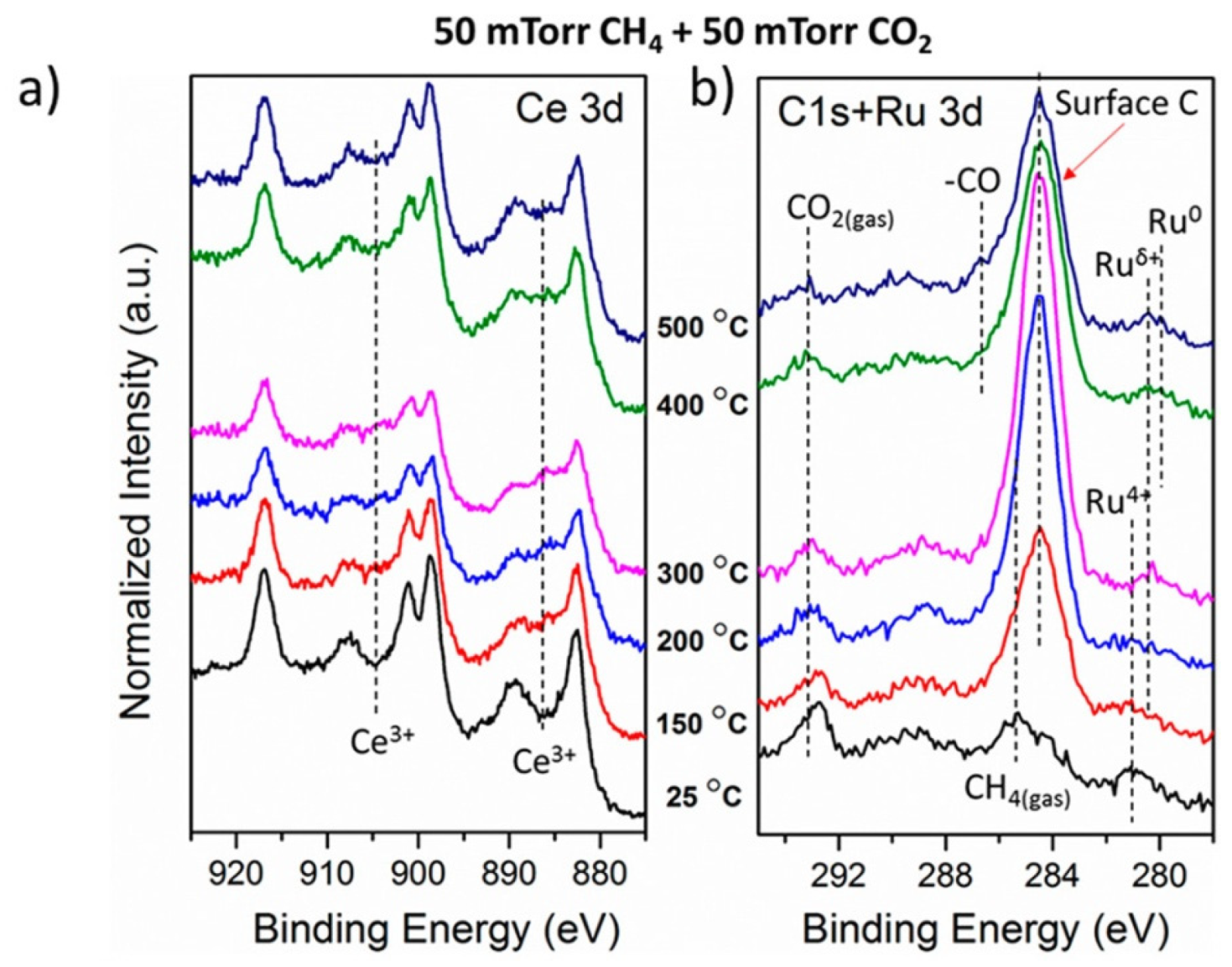

13, 15), suggesting that the active metal sites were largely covered by support, which altered the geometric ensembles and improved the thermal stability of Pd catalysts. For example, the adsorption of H 2 or CO on Pd was extremely suppressed upon the formation of SMSI (refs. 12, 13, 14, SMSI has been widely exploited to tune catalytic performances of group VIII NPs by engineering geometric and/or electronic structures of these metal sites. Since the very first discovery of SMSI by Tauster et al. Such an interface provides synergistic properties to regulate catalysis by modifying the electronic (charge transfer between the metal sites and the support) and/or geometric (decoration or coverage of metal sites by the support) parameters, and also by modulating the reaction pathways, e.g., lattice oxygen in oxide supports may directly participate in catalytic reactions 7 multicomponent interfaces can enable tandem reaction pathways that do not exist on single-component active sites 8, 9.Īs a classic prototype in metal–support interactions, the strong metal–support interaction (SMSI) has been defined as the encapsulation of NPs, usually group VIII metals, by partially reduced oxide supports during high-temperature hydrogen (H 2) treatment 10, 11. Subsequently, the metal–support interface is constructed. Conventionally, these catalysts are synthesized by anchoring the active metal nanoparticles (NPs) onto certain high-surface-area supports to increase the dispersion of catalytically active sites and stabilize the metal against leaching 5, 6, 7. Supported metal catalysts have long been recognized as the most important group of heterogeneous catalysts for fundamental investigations and modern chemical industries 1, 2, 3, 4. Detailed electron microscopic and spectroscopic experiments coupled with computational modeling demonstrate that the compelling performance stems from the SMSIR, favoring the formation of surface hydrogen on Pd instead of hydride. The final structure, denoted as Pd–Fe 3O 4–H, exhibits excellent catalytic performance in semi-hydrogenation of acetylene with 100% conversion and 85.1% selectivity to ethylene at 80 ☌. Using core–shell nanoparticles (NPs) as a building block, the Pd–FeO x NPs are transformed into a porous yolk–shell structure along with the formation of SMSIR upon treatment under a reductive atmosphere. Here, we demonstrate a strong metal–support interaction via a reverse route (SMSIR) by starting from the final morphology of SMSI (fully-encapsulated core–shell structure) to obtain the intermediate state with desirable exposure of metal sites. Engineering strong metal–support interactions (SMSI) is an effective strategy for tuning structures and performances of supported metal catalysts but induces poor exposure of active sites.
- Home
- Services
- About
- News
- Contact
- Shader model 3-0
- Gta 5 mod menu download xbox one
- Cool og gamertags not taken
- Hr xps peak pd 3d
- Combos de the king of fighters 2002 para maquinitas
- Dragon age inquisition patch 1-12 cheats
- Run 8 train simulator multiplayer servers
- Enochian chess set
- Spartacus all seasons watch online with english subtitles
- Rufus ubuntu
- Ems topgun 3 ps2
- Pharaoh game
- Hex color code soft navy blue
- Concorde c40 modem driver
- Flstudio video
- Convert pdf to cad autocad 2018
- Xbox 1 controller driver
- Pokemon soul silver rom nds file
- Price zimbra network edition
- Bescor mp-101 motorized pan tilt head
- Free mujhse dosti karoge movie online
- Myanmar xxx
- Re download os x el capitan
- Clear cookies and cache in firefox mac os x
- Cant upgrade to 64 bit windows 10
- Ibm i access client solutions screen flashes
- Novel dewasa online
- Mac website builder software
- Download film wiro sableng 2018 bluray
- Seagate file recovery for windows registration key free
- Best tamil gana songs mp3 free download
- Mac battery life short
- Convert mod to avi file
- Intel centrino advanced n 6235 driver windows 7 64 bit
